The early stages of a medical device development project are a time of both opportunity and risk. There is an idea that has the potential to transform health outcomes and create commercial value, but progress can be slow and unpredictable. It can even be challenging knowing how to start, where to allocate limited capital, or what you can do to unblock a stalled project. A prototype sprint approach offers a solution, with a controlled timeline, proven outcomes, and a process designed to mitigate risks.
In this blog, we’ll explain what a prototype sprint process looks like in medical device design, including the key components. We’ll also explore in detail why this approach is beneficial, whether you are a start-up company or an established OEM.
In summary, a prototype sprint approach to medical device design produces tangible outputs and strategic value, both of which help with decision-making, accessing further funding, and progressing your project to more advanced stages of development.
Table of Contents
What Is a Medical Device Prototype Sprint?
A prototype sprint brings structure and defined outcomes to the process of developing a new medical device product. Scope creep is controlled, budgets are set, and timelines are fixed, giving you clarity in terms of what you can expect from the process and what it will produce at the end.
What the prototype sprint process produces is a manufacturable prototype of your medical device product ready for full development.
Key features of a prototype sprint medical device design process include:
- Structured, proven process.
- Led by experienced medical device professionals with buy-in and commitment from key stakeholders on your team.
- Fixed timeline of between 90 and 180 days.
- Fixed scope engagement and clear budget.
- Defined outcomes that include design documentation and a manufacturable prototype.
Stages of a Prototype Sprint
There are six main stages in a prototype sprint medical device design process:
- Requirements stage – defining user requirements, functional requirements, device specifications, and project goals.
- Concept stage – creation of multiple concepts and solutions for analysis and review, with the best options selected for the next stage.
- Development stage – review manufacturing methods and materials for the selected concepts, as well as creating 3D CAD models. Where applicable, concepts are narrowed further based on further analysis and review.
- 1st prototype stage – production of physical models of each prototype for bench testing, evaluation, and review of key performance characteristics. This includes size, fit, and ergonomics.
- Refinement stage – select one design for further refinement, including design for manufacturing and design for assembly considerations.
- 2nd prototype stage – production of a manufacturable prototype.
Advantages of Prototype Sprint Medical Device Design
We have split the advantages of using a prototype sprint process into two categories:
- Tangible outputs – the outcomes you can expect and why those outcomes matter to your business and product development process.
- Strategic value – the strategic value the prototype sprint process adds to your company, project, and medical device product.
Prototype Sprint Benefits: Tangible Outputs
Manufacturable Prototype
A prototype sprint process produces not just a prototype that proves a concept, but a manufacturable prototype, i.e., a prototype that is closer to commercialisation. This distinction is important as the gap between a proof-of-concept prototype and a manufacturable prototype can result in delays of years in medical device design projects.
With a prototype sprint approach, there is a focus from the start on closing the gap between concept and manufacturable prototype. As a result, the process involves reviewing materials, manufacturing methods, and assembly in addition to proving the concept of the device.
Having a manufacturable prototype at the end of the sprint will help put your company in a strong position in your development journey. For example, there are large practical and psychological differences between showing a stakeholder a concept prototype and a manufacturable prototype. One is a promise or hope of something in the future. The other is real-world, tangible evidence of a medical device product.

Regulatory Clarity
Medical device regulations are complex. As a result, the temptation is to delay regulatory thinking until later in the process when ideas and concepts are clearer. This approach often causes delays as a result of design, funding, or regulatory setbacks.
The best approach is to embed regulatory thinking from the start so there are no surprises down the road. This includes making design decisions during the sprint for regulatory purposes as well as performance and manufacturability.
Having regulatory clarity early in the process also helps with understanding the regulatory pathway/s you will need to follow to obtain approval, including the regulatory classification of your device and the testing that will be required.

Defined Cost and Timeline
It is common in the medical device design industry for development projects to be open-ended in terms of scope, timeline, and, often, cost. A tightly structured prototype sprint process with a defined deliverable brings valuable clarity and predictability. Upfront, you will know the budget, endpoint, and output – how much it costs, the timeline involved, and what you will get at the end.
Prototype Sprint Benefits: Strategic Value
Satisfy Existing Investors
The funding of medical device projects is typically structured in stages tied to milestones. The production of a manufacturable prototype is a credible and meaningful milestone in that funding journey.
This gives existing investors confidence not just in the product but in the ability of your team’s execution capabilities in relation to the technical complexities, regulatory challenges, and manufacturing realities of developing a new medical device product. It also demonstrates to investors that you have used early project capital to reach a well-defined and meaningful milestone.
Positions You for Further Investment
Getting from seed and angel funding at the earliest stages of your project to Series A funding can be complex, challenging, and time-consuming. Ideas and concepts are not enough for Series A investors. They want to see risks mitigated and tangible proof of the potential commercialisation of your medical device. The evidence that Series A investors look for includes:
- Evidence the device can be manufactured at scale on a production line.
- Evidence there is a well-understood and clear regulatory pathway.
- Evidence your team is capable of delivering.
The word “evidence” is important here, as you will have documentation and data to back up what you are saying once you have gone through a prototype sprint process.
Early Identification of Technical Risk
One of the key objectives of a prototype sprint process is to surface problems, challenges, and risks as early as possible. This is the time when they are the least costly and time-consuming to fix, address, and mitigate.
This fact is often underestimated in the drive to develop a medical device concept into a product. The reality is that it is exponentially more expensive to correct an issue that is identified at the validation stage or after you have made your regulatory submission. Surfacing problems early is essential.
Builds Momentum
It can be tough to build momentum in the early stages of a medical device development project, especially if you are a start-up company. Technical complexities, regulatory complexities, long (often open-ended) timelines, and capital requirements can result in slow or stalled progress.
Adopting a prototype sprint approach will help you build momentum – real, demonstrable momentum. This keeps morale high, energies focused, and stakeholders on board.
Enhances Confidence
Following on from the previous point about building momentum, developing a manufacturable prototype as part of a controlled but accelerated timeline can enhance confidence in your product and wider business project.
We are not just talking about enthusiasm and energy. Those things are important, but the real value comes from the validated confidence that you get with a product prototype that has been through robust bench testing, design review, regulatory scoping, and manufacturability stages.
Validated confidence in your product will give you information to make decisions on the next stages of the project.
Clarity of IP Position
Intellectual Property (IP) is critical to the commercial value of your medical device product. A well-structured prototype sprint process will surface important questions about your IP. Examples include:
- What can be protected?
- What IP already exists?
- What design decisions could create freedom-to-operate issues?
The last point is worth expanding on further – freedom-to-operate (FTO). IP considerations are not just about patenting your own idea. It’s also about making sure your product doesn’t infringe on an existing patent. This can arise even in situations where you have a genuinely novel device, as something within the design (often arrived at inadvertently) could infringe on an existing patent that would impact your ability to manufacture or sell the product.
A prototype sprint process doesn’t include an FTO analysis – that is the job of experienced patent lawyers and attorneys. However, the prototype sprint process will produce a documented record of design decisions that facilitates early discussions on IP and FTO with your legal representation.
Strong Relationship with a Trusted Manufacturing Partner
Engaging in a prototype sprint process with a company like Arrotek develops strong relationships, mutual understanding, and knowledge that can be utilized as your project moves through the latter stages, up to and including scale-up manufacturing.
The Starting Point Changes Everything
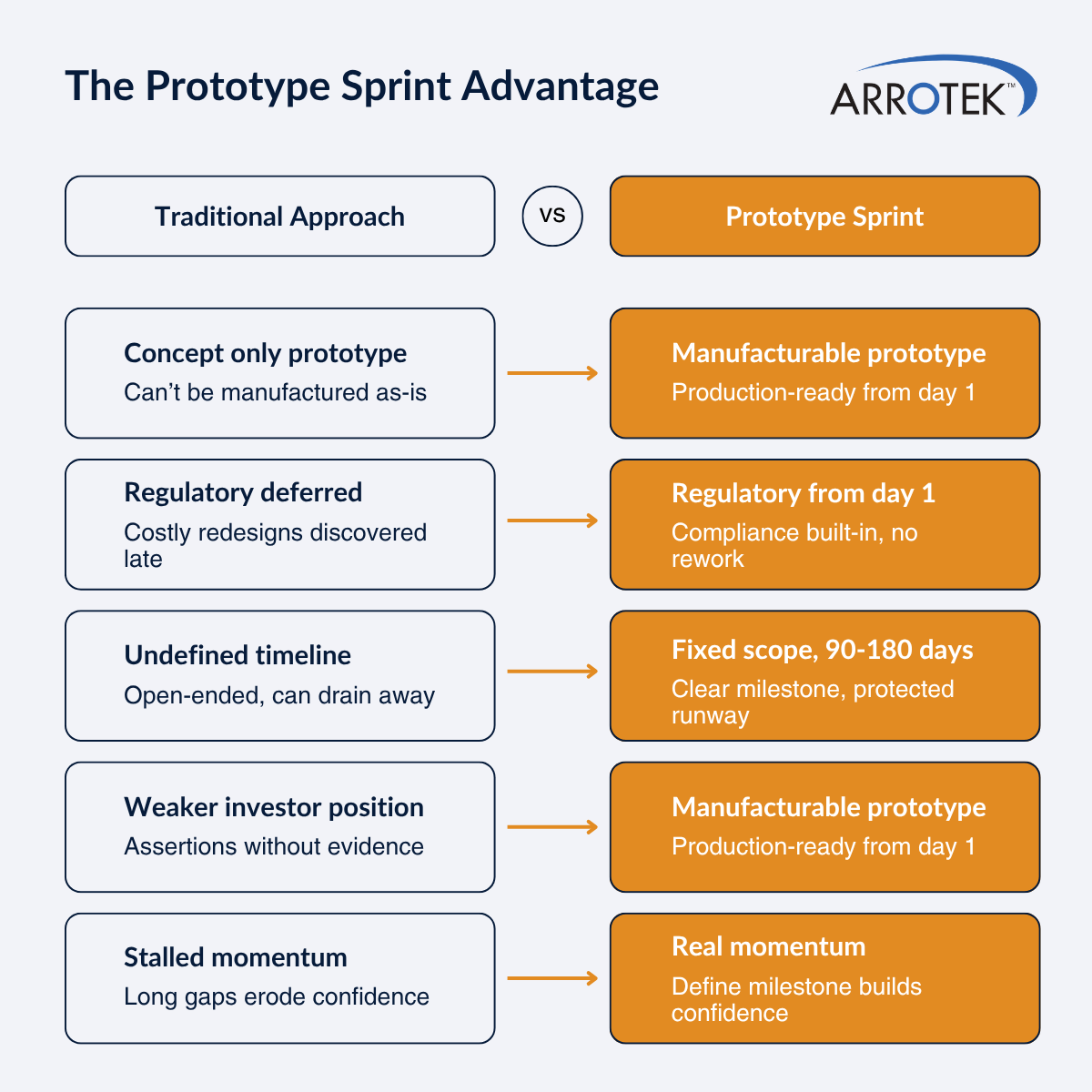
It’s clear in this blog that Arrotek offers a Prototype Sprint package to medical device start-ups and established OEMs. However, the sprint concept is much more than a service offered by a potential partner. For your business, it’s a strategic decision. Do you move forward using the traditional open-ended approach? Or do you adopt a structured, controlled, and measured sprint process that mitigates risks, removes uncertainty, and produces tangible outcomes?
Are you ready to sprint? We’d love to explore whether the prototype sprint option is right for your project or medical device idea. Get in touch to start a conversation with one of our consultants.





