Medical Device Design Services
Expert design and development services for minimally invasive medical devices, from early concept through to prototype production, regulatory submission, and scale-up manufacturing.
Specialising in interventional catheters, guidewires, specialty needles, and minimally invasive access devices.
A Specialist Partner for Complex Medical Device Projects
We work with OEMs, MedTech startups, and established medical device companies to move complex device concepts from idea to validated, manufacturing-ready designs, while reducing development risk and avoiding costly redesigns.
Our focus is on precision, minimally invasive devices. This includes catheter systems, guidewires, specialty needles, and access devices that require exacting engineering and deep regulatory knowledge to bring to market. This narrow focus means our team has accumulated significant, repeatable expertise in the specific engineering challenges these devices present.
We offer a fully integrated service from concept to commercialisation, covering design, prototyping, regulatory documentation, and DFM (design for manufacturing), and pilot builds.
What Our Clients Say

I know the team at Arrotek is thorough, trustworthy, and efficient. Arrotek had everything we needed including the right skillset as well as an established organization ready to hit the ground running with R&D, regulatory submission preparation, production, etc.

Arrotek really became an extension of our R&D and product development efforts and strategy. We don't have to worry about the catheter other than providing input to the Arrotek team. We can have that peace of mind to completely outsource that to Arrotek.
Medical Device Design Capabilities
Our design and development team has deep experience across a focused range of minimally invasive device categories: catheter, needle, guidewire, and other minimally invasive devices.
Catheter Design and Development
We design and develop a full range of advanced catheter technologies, including:
- Thin-wall catheters
- Reinforced catheters, including braided and coiled catheters
- Balloon catheters
- Steerable catheters
- Ablation catheters
- Multi-lumen catheters
- Diagnostic catheters
Catheter design is one of the most technically demanding areas of medical device engineering. The performance of a catheter system (its pushability, trackability, torque response, tip behavior, etc) depends on a precise combination of material selection, construction method, and geometry. Small changes to wall thickness, braid angle, or durometer profile can have significant effects on clinical performance and manufacturability.
Our catheter design team works across the full range of construction techniques: extruded single-lumen and multi-lumen shafts, braided and coiled reinforcement, heat-bonded and reflowed tip assemblies, and over-molded handle and hub configurations.
We select materials and construction approaches based on the specific clinical requirements of each device. This could mean, for example, prioritizing flexibility for navigation through tortuous anatomy, or stiffness for precise force transmission in interventional procedures.
Because our design team works alongside our manufacturing operation, catheter designs are engineered for production from the outset. Prototype builds are completed in-house, allowing for rapid iteration and direct feedback between design and manufacturing engineers. This compresses development timelines and reduces the risk of costly design changes at transfer to production.
Specialty Medical Needle Design
We design specialty needles for interventional, biopsy, drug delivery, and access applications, including:
- Micro needle design for minimally invasive drug delivery and sampling
- Cannula and stylet design for access and drainage systems
- Custom tip geometry and grind profiles for specific clinical requirements
Specialty needle design requires precision at a scale that leaves little margin for error. Tip geometry, wall configuration, material hardness, and surface treatment all directly impact the clinical performance of the device. For example, clinical performance in relation to penetration force, core retention, flow characteristics, or patient comfort.
Getting these parameters right demands both engineering rigor and deep familiarity with the clinical context in which the device will be used.
Our needle design capability is closely integrated with our manufacturing expertise in precision metalworking. This means we can evaluate the manufacturability of a tip geometry or wall specification during the design phase, before a prototype is ever built. We can then advise on whether a given design can be produced reliably at the required tolerances and volumes.
We regularly work on needle designs that require custom grind profiles, tight-tolerance thin-wall constructions, or specialist coatings such as hydrophilic, PTFE, and silicone formulations. We bring that same manufacturing knowledge to projects from the earliest stages of the design process.
Guidewire Design
We design guidewires for vascular, neurovascular, and interventional applications, from standard access wires to high-performance coronary and neurovascular constructions.
Our guidewire design capability covers core wire selection, tip shaping, coating specification, and torque performance optimisation across a range of diameters and stiffness profiles. We work across standard, hydrophilic, and nitinol-based constructions.
Guidewire performance is defined by the relationship between stiffness, flexibility, and torque response along the length of the wire. These properties must be engineered simultaneously as they can interact in ways that are not always intuitive.
A guidewire that tracks well through a straight vessel, for example, may perform poorly in a tortuous anatomy. A wire optimized for torque transmission, on the other hand, may sacrifice the tip flexibility needed for atraumatic navigation.
Balancing these properties for a specific clinical application is the central engineering challenge in guidewire design.
Our guidewire design work covers the full range of clinical applications, from standard vascular access wires to high-performance neurovascular and coronary guidewires where the performance envelope is particularly demanding.
We specify core wire materials and temper profiles, design tip shaping and forming processes, and select and validate coatings for lubricity, durability, and biocompatibility. We also evaluate torque performance through bench testing against clinically relevant use conditions.
Where nitinol is specified (for example, for its superelastic properties in shapeable or steerable tip designs), we bring specific experience in nitinol processing and heat treatment to the design process.
Minimally Invasive and Interventional Device Design
Beyond catheters and needles, our team designs a range of interventional devices for vascular access, neurovascular intervention, interventional oncology, and interventional radiology applications. We are experienced in the specific regulatory pathways, material requirements, and clinical use conditions these devices face.
Minimally invasive devices span a broad and technically diverse product landscape. That product landscape includes everything from introducers and sheaths to dilators, snares, retrieval devices, and combination systems that integrate multiple functional elements into a single deliverable construct.
What these devices share is a requirement for precise engineering under tight dimensional constraints, combined with a deep understanding of the clinical environment in which they operate.
Our team approaches each interventional device design with the clinical use case as the starting point. We consider how the device will be positioned, what forces it will experience, how it will interact with anatomy and with other devices in the procedural stack, and what failure modes look like in a worst-case clinical scenario.
This clinical thinking shapes material selection, geometry, and construction choices from the earliest concept stage. Our approach is also integrated with the regulatory and risk management disciplines that minimally invasive devices require.
The result is a design process that produces devices that are not only engineered to perform, but documented and validated to support regulatory submission.
Medical Specialties We Serve
Our device design work spans the following clinical areas:
Within each specialty, we understand the clinical workflow, device handling requirements, and regulatory classification landscape that shapes design decisions. This means we can contribute meaningfully from the earliest stages of concept development.
| Clinical Area | Specialty |
|---|---|
| Interventional Cardiology | Treatment and diagnosis of conditions in the heart and blood vessels |
| Interventional Radiology | For the diagnosis of conditions and/or to guide treatments |
| Interventional Oncology | For the diagnosis and treatment of cancer and related problems |
| Neurovascular | For the diagnosis and treatment of conditions in the blood vessels of the brain, spine, or spinal cord |
| Other | Vascular access, pharma, pain management, spine and biologics, sports medicine |
Our Six-Step Medical Device Design Process
We follow a structured, stage-gated development process aligned to ISO 13485 requirements and FDA design controls (21 CFR Part 820). This approach ensures each project progresses with appropriate documentation, risk management, and design verification at every stage.
Each stage ends with a formal gate review. Decisions to proceed, iterate, or redirect are made on evidence rather than assumptions, keeping projects on time and within budget.
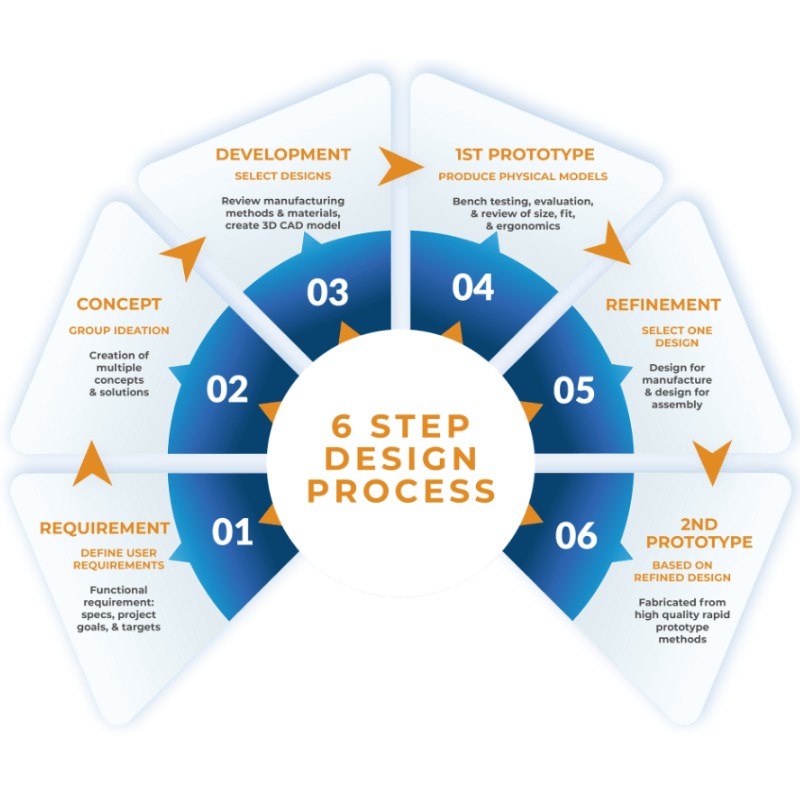
| 1 | Requirements | We define the clinical and functional requirements for the device. This includes what it must do, in what environment, and for which patient population. |
| 2 | Concept Development | Multiple design concepts are generated and evaluated against the design inputs. We apply engineering analysis, material shortlisting, and manufacturability assessments to narrow down to the most viable approaches. This validates feasibility before committing budget to detailed design. |
| 3 | Detailed Development | We further review manufacturing methods and materials, and 3D CAD models are produced. Concept options are narrowed further. |
| 4 | 1st Prototype | Physical models of each concept are produced for bench testing and further assessment. This also helps reduce design risks before regulatory submissions. |
| 5 | Refinement | One design is selected. It goes through further refinement, including design for manufacturability (DFM). |
| 6 | 2nd Prototype | A manufacturable prototype is produced. |

Regulatory Expertise Built Into the Design Process
Regulatory compliance is not a downstream activity at Arrotek. Instead, we integrate compliance consideration into every stage of the design process. Our team is experienced in the requirements of all major regulatory frameworks applicable to minimally invasive medical devices:
- ISO 13485:2016: Our quality management system governs all design and development activities
- FDA 21 CFR Part 820: Design Controls requirements are followed throughout the development process
- EU MDR (Regulation 2017/745): We design with European market requirements in mind, including technical file structure and clinical evidence requirements
- IEC 62366: Human Factors Engineering and usability principles are applied throughout design
- ISO 10993: Biocompatibility risk management for materials selection and testing strategy
- Risk management per ISO 14971: DFMEA and hazard analysis are part of our standard design process
This regulatory fluency means we can advise on classification, predict the documentation requirements your project will face, and structure design outputs from day one to support your submission, whether that is a 510(k), CE marking under MDR, or other requirement.
Design for Manufacturing
Engineering medical device products that can be commercially produced
A medical device that works in the lab but cannot be manufactured reliably at scale is not a finished product. At Arrotek, DFM (Design for Manufacture) principles are applied throughout the design process.
Because our design team works directly alongside our manufacturing capability, we make material and geometry decisions with production in mind from the outset. This means:
- Tighter alignment between prototype and production-intent design
- Fewer design changes required at transfer to manufacturing
- Better visibility of target manufacturing costs during the design phase
- Reduced risk of scale-up delays caused by unmanufacturable features
For clients with their own manufacturing operations, we can document DFM outputs in a format that transfers cleanly to your own production team or CMO.

Why Work With Arrotek
Focused Expertise
We work exclusively on minimally invasive, catheter-based, and needle-based devices. This focus means our engineering team has deep, repeatable experience with the specific challenges these products present.
Design & Manufacturing
Our design capability sits alongside our manufacturing operation. This supports design for manufacturing engineering and allows prototypes to be built, tested, and iterated quickly, without the need for third-party suppliers.
Full Lifecycle Support
We support the full product lifecycle, from initial concept design to prototype development to regulatory support to scale-up manufacturing to ongoing product version iteration and development.
Flexible Engagement Model
We will adapt our engagement model to your situation, e.g., augmenting your existing engineering team on a specific workstream, taking full ownership of a development project, or providing targeted, specialized support.
Intellectual Property Protection
All client work is treated in strict confidence, and you will retain 100% of the intellectual property rights over your device concept and design outputs. We do not use client IP for any purpose beyond the agreed scope of work.
Proven Track Record
We have a proven track record of successful projects designing and developing novel medical device products and components for medical entrepreneurs, start-up companies, and established OEMs.
Who We Work With
Medical Device OEMs
Established OEMs work with Arrotek to extend their internal engineering capacity, accelerate specific development programs, or access specialist capabilities such as catheter design or micro needle engineering. We have experience working within existing quality systems and adapting our outputs to fit your DHF structure and document control requirements.
Start-Ups & Early Stage Companies
Early-stage companies and entrepreneurs use Arrotek to access the engineering and regulatory expertise needed to take a device concept from idea to investable prototype. We are experienced in helping founders understand the regulatory pathway for their device, structure their development program efficiently, and produce manufacturable prototypes for testing.
Client Video Testimonial
Dr. Habib Frost
CEO of Neurescue
Quote from the video: “Whether its in manufacturing, development, or regulatory – we’ve achieved stellar results.”
Discuss Your Medical Device Project
Share your device concept or development challenge and our engineering team will review your requirements and advise on feasibility, regulatory considerations, and next steps.
You can submit your enquiry via the form or email us at [email protected]. All inquiries are treated in strict confidence.
We support medical device companies across the USA, Europe, and internationally, from early-stage concepts through to scalable manufacturing. There is no minimum project size required to begin a technical review.
Frequently Asked Questions
What types of medical devices does Arrotek design?
Arrotek specializes in minimally invasive medical devices, with particular expertise in interventional catheters, guidewires, specialty needles, cannulas, and access devices. We work across interventional cardiology, interventional radiology, interventional oncology, neurovascular, and vascular access applications.
What is the typical timeline for a medical device design project?
Timelines depend heavily on device complexity, regulatory classification, and how many development iterations are required. A typical concept-to-functional-prototype program for a catheter-based device runs between four and twelve months. We provide a project plan and timeline estimate during initial scoping.
Do you work with startups as well as established companies?
Yes. We work with early-stage companies and entrepreneurs who need to develop a first prototype and understand their regulatory pathway, as well as established OEMs who need to extend their engineering capacity or access specialist capabilities.
What regulatory standards do you work to?
Our quality management system is ISO 13485 certified. Our design process follows FDA 21 CFR Part 820 Design Controls requirements and EU MDR technical documentation requirements. We apply ISO 14971 risk management and IEC 62366 human factors principles as standard throughout the design process.
Do you handle the full project or can you support just part of it?
Both. We can take full ownership of a design and development program from concept to manufacturing transfer, or we can provide targeted support on a specific phase (such as detailed design, prototype build, or DFM review). We’ll adapt our involvement to your needs and existing team structure.
Where is Arrotek based?
Our medical device design teams are based in Massachusetts, USA and Sligo, Ireland. We also have manufacturing facilities in Massachusetts and Sligo, as well as in Costa Rica.